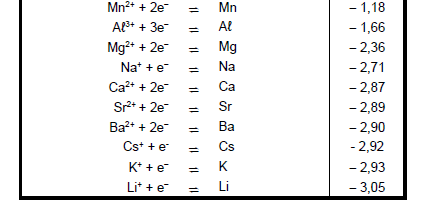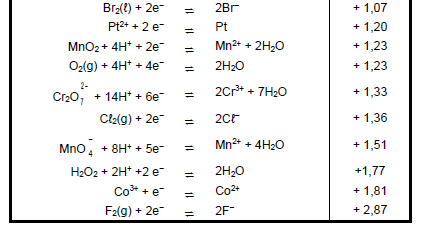PHYSICAL SCIENCES PAPER 2 GRADE 12 QUESTIONS - NSC EXAMS PAST PAPERS AND MEMOS NOVEMBER 2020
Share via Whatsapp Join our WhatsApp Group Join our Telegram GroupPHYSICAL SCIENCES: CHEMISTRY (PAPER 2)
GRADE 12
NATIONAL SENIOR CERTIFICATE
NOVEMBER 2020
INSTRUCTIONS AND INFORMATION
- Write your examination number and centre number in the appropriate spaces on the ANSWER BOOK.
- This question paper consists of TEN questions. Answer ALL the questions in the ANSWER BOOK.
- Start EACH question on a NEW page in the ANSWER BOOK.
- Number the answers correctly according to the numbering system used in this question paper.
- Leave ONE line between two subquestions, e.g. between QUESTION 2.1 and QUESTION 2.2.
- You may use a non-programmable calculator.
- You may use appropriate mathematical instruments.
- Show ALL formulae and substitutions in ALL calculations.
- Round off your FINAL numerical answers to a minimum of TWO decimal places.
- Give brief motivations, discussions, etc. where required.
- You are advised to use the attached DATA SHEETS.
- Write neatly and legibly.
QUESTION 1: MULTIPLE-CHOICE QUESTIONS
Various options are provided as possible answers to the following questions. Choose the answer and write only the letter (A–D) next to the question numbers (1.1 to 1.10) in the ANSWER BOOK, e.g. 1.11 E.
1.1 Which ONE of the following is the general formula for the alkanes?
- CnH2n
- CnH2n-2
- CnH2n+2
- CnH2n+2O (2)
1.2 The EMPIRICAL FORMULA of hexanoic acid is …
- C3H6O2
- C6H6O2
- C6H12O2
- C3H6O (2)
1.3 Which ONE of the following is the CORRECT structural formula for METHYL ETHANOATE?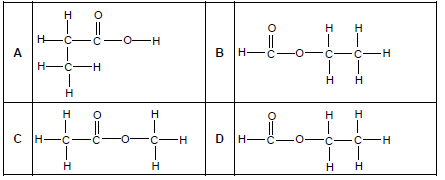
1.4 Zinc (Zn) granules react as follows with EXCESS hydrochloric acid solution, HCℓ(aq):
Zn(s) + HCℓ(aq) → ZnCℓ2(aq) + H2(g)
Which ONE of the following combinations of volume and concentration of HCℓ(aq) will result in the highest INITIAL reaction rate for the same mass of zinc granules used? (Assume that the zinc granules are completely covered by the acid in all cases.)
| VOLUME HCℓ(aq) (cm3) | CONCENTRATION HCℓ(aq) (mol∙dm-3) | |
| A | 50 | 0,5 |
| B | 100 | 1,0 |
| C | 200 | 0,1 |
| D | 200 | 0,5 |
(2)
1.5 The role of a catalyst in a chemical reaction is to increase the …
- yield.
- activation energy.
- heat of reaction.
- rate of the reaction. (2)
1.6 Consider the equilibrium represented by the balanced equation below:
2CrO2-4 (aq) + 2H+(aq) ⇌ Cr2O2-7(aq) + H2O(ℓ) ΔH < 0
Which ONE of the following changes to the equilibrium will favour the forward reaction?
| TEMPERATURE | pH | |
| A | Decrease | Increase |
| B | Decrease | Decrease |
| C | Increase | Increase |
| D | Increase | Decrease |
(2)
1.7 The conjugate base of HPO2-4 is …
- OH-
- PO3-4
- H2PO-4
- H3PO4 (2)
1.8 Which ONE of the following reactions will proceed spontaneously under standard conditions?
- Ni2+(aq) + H2(g) → Ni(s) + 2H+(aq)
- Br2(ℓ) + 2Cℓ-(aq) → 2Br- (aq) + Cℓ2(g)
- 2Fe3+(aq) + 2I-(aq) → 2Fe2+(aq) + I2(s)
- 2Cu+(aq) + Pb2+(aq) → 2Cu2+(aq) + Pb(s) (2)
1.9 The simplified diagram below represents an electrochemical cell used for the PURIFICATION of copper.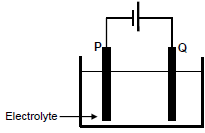
Which ONE of the graphs below represents the CHANGE IN MASS of electrodes P and Q during the purification process? (2)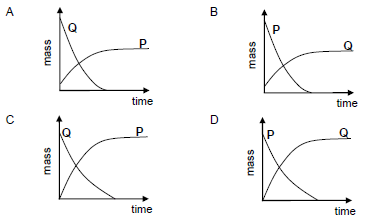
1.10 Eutrophication in water is caused by …
- algal bloom.
- bacterial nitrogen fixation.
- an increase in plant nutrients.
- a depletion of oxygen concentration. (2)
[20]
QUESTION 2 (Start on a new page.)
The letters A to E in the table below represent five organic compounds.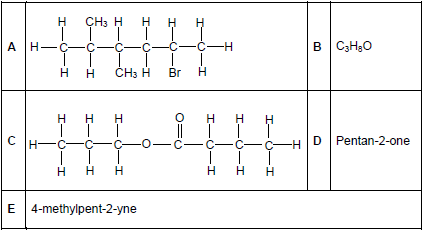
Use the information in the table to answer the questions that follow.
2.1 For compound D, write down the:
2.1.1 Homologous series to which it belongs (1)
2.1.2 IUPAC name of a FUNCTIONAL ISOMER (2)
2.2 Write down the:
2.2.1 IUPAC name of compound A (3)
2.2.2 STRUCTURAL FORMULA of compound E (2)
2.3 Compound B is a primary alcohol.
2.3.1 Write down the meaning of the term primary alcohol. (2)
Compound B reacts with another organic compound X to form compound C.
Write down the:
2.3.2 Type of reaction that takes place (1)
2.3.3 IUPAC name of compound X (1)
[12]
QUESTION 3 (Start on a new page.)
The relationship between boiling point and the number of carbon atoms in straight chain molecules of aldehydes, alkanes and primary alcohols is investigated. Curves A, B and C are obtained.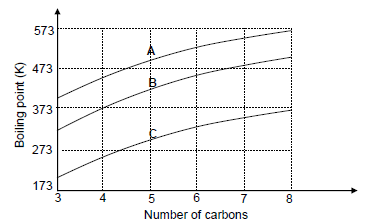
3.1 Define the term boiling point. (2)
3.2 Write down the STRUCTURAL FORMULA of the functional group of the aldehydes. (1)
3.3 The graph shows that the boiling points increase as the number of carbon atoms increases. Fully explain this trend. (3)
3.4 Identify the curve (A, B or C) that represents the following:
3.4.1 Compounds with London forces only (1)
3.4.2 The aldehydes
Explain the answer. (4)
3.5 Use the information in the graph and write down the IUPAC name of the compound with a boiling point of 373 K. (2)
3.6 Write down the IUPAC name of the compound containing five carbon atoms, which has the lowest vapour pressure at a given temperature. (2)
[15]
QUESTION 4 (Start on a new page.)
The flow diagram below shows how various organic compounds can be prepared using compound P as starting reagent.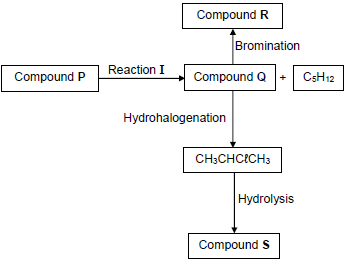
4.1 Write down the meaning of the term hydrohalogenation.(2)
4.2 Write down the STRUCTURAL FORMULA of compound Q. (2)
4.3 Reaction I is an elimination reaction.
Write down the:
4.3.1 TYPE of elimination reaction (1)
4.3.2 MOLECULAR FORMULA of compound P (1)
4.4 Write down the IUPAC name of compound R. (2)
4.5 For the HYDROLYSIS REACTION, write down the:
4.5.1 Balanced equation using structural formulae (5)
4.5.2 TWO reaction conditions (2)
[15]
QUESTION 5 (Start on a new page.)
The reaction of calcium carbonate (CaCO3) and EXCESS dilute hydrochloric acid (HCℓ) is used to investigate one of the factors that affects reaction rate. The balanced equation for the reaction is:
CaCO3(s) + 2HCℓ(aq) → CaCℓ2(aq) + H2O(ℓ) + CO2(g)
The same mass of CaCO3 is used in all the experiments and the temperature of the hydrochloric acid in all experiments is 40 °C.
The reaction conditions for each experiment are summarised in the table below.
| EXPERIMENT | VOLUME OF HCℓ(aq)(cm3) | CONCENTRATION OF HCℓ(aq)(mol∙dm-3) | STATE OF DIVISION OF CaCO3 |
| A | 500 | 0,1 | granules |
| B | 500 | 0,1 | lumps |
| C | 500 | 0,1 | powder |
5.1 For this investigation write down the:
5.1.1 Dependent variable (1)
5.1.2 Independent variable (1)
The carbon dioxide gas, CO2(g), produced during EXPERIMENT A, is collected in a gas syringe. The volume of gas collected is measured every 20 s and the results obtained are shown in the graph below.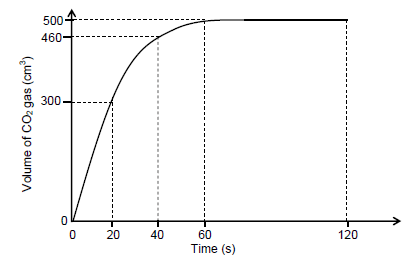
5.2 What can be deduced from the graph regarding the RATE OF THE REACTION during the time interval:
5.2.1 20 s to 40 s (1)
5.2.2 60 s to 120 s (1)
5.3 Calculate the average rate (in cm3∙s-1) at which CO2(g) is produced in the experiment. (3)
5.4 How will the volume of CO2(g) produced in experiment B compare to that produced in experiment A? Choose from GREATER THAN, SMALLER THAN or EQUAL TO. (1)
5.5 A graph is now drawn for experiment C on the same set of axes. How will the gradient of this graph compare to the gradient of the graph for experiment A? Choose from GREATER THAN, SMALLER THAN or EQUAL TO.
Use the collision theory to fully explain the answer. (4)
5.6 Assume that the molar gas volume at 40 °C is 25,7 dm3∙mol-1. Calculate the mass of CaCO3(s) used in experiment A. (4)
[16]
QUESTION 6 (Start on a new page.)
The dissociation of iodine molecules to iodine atoms (I) is a reversible reaction taking place in a sealed container at 727 °C. The balanced equation for the reaction is: I2(g) ⇌ 2I(g)
Kc for the reaction at 727 °C is 3,76 x 10-3.
6.1 Write down the meaning of the term reversible reaction. (1)
6.2 At equilibrium the pressure of the system is increased by decreasing the volume of the container at constant temperature.
How will EACH of the following be affected? Choose from INCREASES, DECREASES or REMAINS THE SAME.
6.2.1 The value of the equilibrium constant (1)
6.2.2 The number of I2 molecules (1)
6.3 Explain the answer to QUESTION 6.2.2 by referring to Le Chatelier's principle.(2)
6.4 At 227 °C, the KC value for the reaction above is 5,6 x 10-12.
Is the forward reaction ENDOTHERMIC or EXOTHERMIC?
Fully explain the answer. (4)
6.5 A certain mass of iodine molecules (I2) is sealed in a 12,3 dm3 flask at a temperature of 727 °C (Kc = 3,76 x 10-3).
When equilibrium is reached, the concentration of the iodine atoms is found to be 4,79 x 10-3 mol∙dm-3. Calculate the INITIAL MASS of the iodine molecules in the flask. (9)
[18]
QUESTION 7 (Start on a new page.)
7.1 Ethanoic acid (CH3COOH) is an ingredient of household vinegar.
7.1.1 Is ethanoic acid a WEAK acid or a STRONG acid? Give a reason for the answer. (2)
7.1.2 An ethanoic acid solution has a pH of 3,85 at 25 °C. Calculate the concentration of the hydronium ions, H3O+(aq), in the solution. (3)
Sodium ethanoate, CH3COONa(aq), forms when ethanoic acid reacts with sodium hydroxide.
7.1.3 Will the pH of a sodium ethanoate solution be GREATER THAN 7, LESS THAN 7 or EQUAL TO 7? (1)
7.1.4 Explain the answer to QUESTION 7.1.3 with the aid of a balanced chemical equation. (3)
7.2 Household vinegar contains 4,52% ethanoic acid, CH3COOH by volume.
A 1,2 g impure sample of calcium carbonate (CaCO3) is added to 25 cm3 household vinegar.
On completion of the reaction, the EXCESS ethanoic acid in the household vinegar is neutralised by 14,5 cm3 of a sodium hydroxide solution of concentration 1 mol∙dm-3. The balanced equation for the reaction is:
CH3COOH(aq) + NaOH(aq) → CH3COONa(aq) + H2O(ℓ)
7.2.1 Calculate the number of moles of the unreacted ethanoic acid. (3)
7.2.2 Calcium carbonate reacts with ethanoic acid according to the following balanced equation:
CaCO3(s) + 2CH3COOH(aq) → (CH3COO)2Ca(aq) + H2O + CO2(g)
Calculate the percentage calcium carbonate in the impure sample if 1 cm3 of household vinegar has a mass of 1 g. (8)
[20]
QUESTION 8 (Start on a new page.)
The electrochemical cell illustrated below is set up under standard conditions.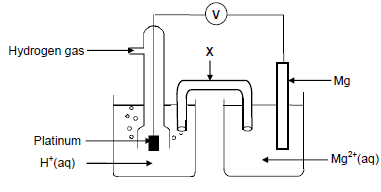
8.1 Component X completes the circuit in the cell. State ONE other function of component X. (1)
8.2 Define the term anode. (2)
8.3 Identify the anode in the cell above. (1)
8.4 Write down the:
8.4.1 Reduction half-reaction that takes place in this cell (2)
8.4.2 NAME or FORMULA of the reducing agent in this cell (1)
8.5 Calculate the initial voltmeter reading of this cell under standard conditions. (4)
8.6 The Mg|Mg2+ half-cell is now replaced by a Cu|Cu2+ half-cell. It is found that the direction of electron flow changes.
Fully explain why there is a change in direction of electron flow by referring to the relative strengths of the reducing agents involved. (3)
[14]
QUESTION 9 (Start on a new page.)
The simplified diagram below represents an electrolytic cell used to electroplate a copper (Cu) coin with silver (Ag).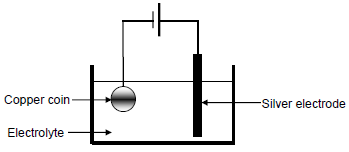
9.1 Define the term electrolysis. (2)
9.2 Which component in the diagram indicates that this is an electrolytic cell? (1)
9.3 Write down the NAME or FORMULA of the electrolyte. (1)
9.4 How will the concentration of the electrolyte change during electroplating? Choose from INCREASES, DECREASES or REMAINS THE SAME.
Give a reason for the answer. (2)
9.5 Write down the balanced equation of the half-reaction that takes place at the silver electrode. (2)
[8]
QUESTION 10 (Start on a new page.)
10.1 The flow diagram below shows how fertiliser B is produced in industry.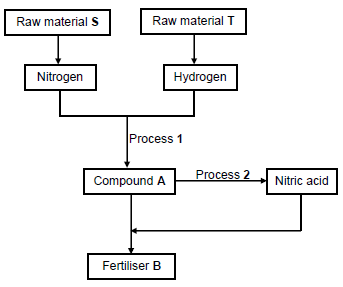
Write down the:
10.1.1 NAME of S (1)
10.1.2 NAME of T (1)
10.1.3 NAME or FORMULA of the catalyst used in process 1 (1)
10.1.4 NAME or FORMULA of compound A (1)
10.1.5 NAME of process 2 (1)
10.1.6 Balanced equation for the formation of fertiliser B (3)
10.2 A 20 kg bag of fertiliser is labelled as follows: 2 : 4 : 3 (X).
10.2.1 What does the ratio on the label represent? (1)
10.2.2 The bag contains 2,315 kg phosphorous.
Calculate the value of X. (3)
[12]
TOTAL: 150
DATA FOR PHYSICAL SCIENCES GRADE 12 PAPER 2 (CHEMISTRY)
TABLE 1: PHYSICAL CONSTANTS
| NAME | SYMBOL | VALUE |
| Standard pressure | pθ | 1,013 x 105 Pa |
| Molar gas volume at STP | Vm | 22,4 dm3∙mol-1 |
| Standard temperature | Tθ | Tθ 273 K |
| Charge on electron | e | e -1,6 x 10-19 C |
| Avogadro’s constant | NA | NA 6,02 x 1023 mol-1 |
TABLE 2: FORMULAE
n = m | n = N NA |
| c = n V or c = m MV | n = V Vm |
| caVa= na cbVb nb | pH= -log[H3O+] |
| Kw = [H3O+][OH-] = 1x10-14 at 298K | |
| Eθ cell = Eθ cathode – Eθ anode Eθ cell = Eθ reduction – Eθ oxidation Eθ cell = Eθ oxidising agent – Eθ reducing agent |
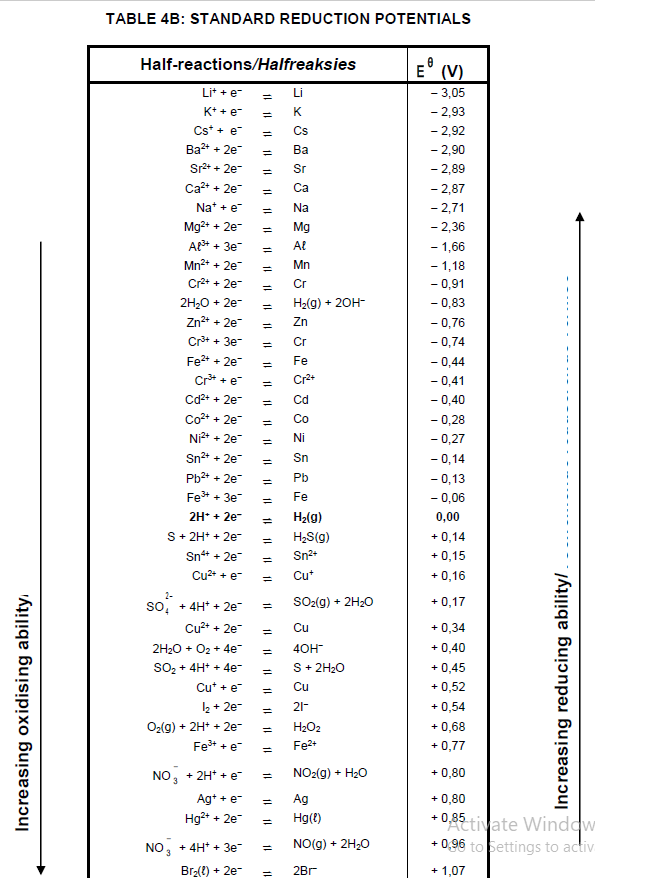
TABLE 4A: STANDARD REDUCTION POTENTIALS