CHEMICAL EQUILIBRIUM QUESTIONS AND ANSWERS GRADE 12
Share via Whatsapp Join our WhatsApp Group Join our Telegram GroupActivity 1
Write the name of a reaction in which all reactants and products are in the same phase (1) [1]
Solution
- Homogeneous ✓ [1]
Activity 2
Consider the following reversible reaction that is in equilibrium in a closed system (the symbols represent chemicals which are unnamed; if you see symbols like X or Y which aren’t on the Periodic Table, it often means you’ve got to work out what kinds of substances they are).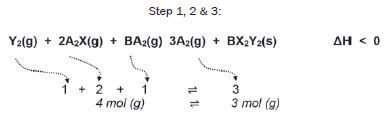
Step 4/5:
∆H < 0
- the forward reaction is exothermic
- the reverse reaction is endothermic
In each of the following cases:
- Explain how the equilibrium will be affected (which reaction will be favoured)
- State how the concentration of the product A2(g) will change.
CASE 1 The reaction system is heated. (3) [3]
Solution
Case 1.
If the reaction system is heated, the temperature of the system increases.
According to Le Châtelier’s Principle, increasing the temperature of the reaction system (i.e. heating it) favours the reaction that will decrease the temperature of the system
- ∴favours the endothermic ✓ reaction but ∆H < 0
- ∴the reverse reaction is endothermic
- ∴the reverse reaction is favoured✓∴the 3A2(g) + BX2Y2(s) reacts faster than it is produced
- ∴the concentration of the A2(g) decreases. ✓
(Remember: The opposite is true if the reaction vessel is cooled.) [3]
hint
You must be able to use Le Châtelier’s Principle to identify the factor that will increase the yield of the products and what influence that factor will have on the reaction AT EQUILIBRIUM
CASE 2 Y2(g) is added to the vessel at constant pressure and temperature (3) [3]
Solution
Case 2.
Adding Y2(g) increases the concentration of the Y2(g). ✓
According to Le Châtelier’s Principle, increasing the concentrationof Y2(g) favours the reaction that decreases the concentration of Y2(g)
- ∴the reaction that uses of Y2(g) as a reactant
- the forward reaction is favoured. ✓
- ∴the rate at which A2(g) and BX2Y2(s) is produced, increases
- the concentration of the A2(g) increases. ✓ [3]
CASE 3 A2(g) is removed from the vessel at constant pressure and temperature. (3) [3]
Solution
Case 3.
Removing A2(g) decreases the concentration of the A2(g). ✓
According to Le Châtelier’s Principle, decreasing theconcentration of the A2(g) favours the reaction that increases the concentration of the A2(g);
- the reaction that makes A2(g) as a product
∴ the forward reaction is favoured. ✓
the rate at which A2(g) and BX2Y2(s) is produced, increases
the concentration of the A2(g) increases. ✓ [3]
CASE 4 Some of the BX2Y2(s) is removed from the system at constant pressure and temperature. (3) [3]
Solution
Case 4.
Removing BX2Y2(s) has no effect on the equilibrium as it is a solid. ✓
Removing a solid from an equilibrium system does not disturb the concentration. ✓ (Solids in equilibrium reactions have very little change in mass, hence they are ignored)
The concentration of A2(g) remain the same. ✓ [3]
CASE 5 The pressure on the system is increased (or the volume is decreased) (3) [3]
Solution
Case 5.
According to Le Châtelier’s Principle, increasing the pressure (or decreasing the volume) of a gaseous system favours the reaction that decreases the pressure of the system by decreasing the total number of gaseous moles in the system. ✓
- the forward reaction is favoured. ✓
- the rate at which A2(g) and BX2Y2(s) is produced, increases
- the concentration of the A2(g) increases. ✓ [3]
hint
There are 4 gaseous moles on the left, and 3 gaseous moles on the right.
Activity 3
Say whether the statement is TRUE or FALSE: If the equilibrium constant for the reaction A2(g) + B2(g) ⇌ 2AB(g) is equal to K, then the equilibrium constant for the reverse reaction
2AB(g) ⇌ A2(g) + B2(g) is also equal to K. (3) [3]
Solution
FALSE✓✓ . ... for the reverse reaction 2AB(g) ⇌ A2(g) + B2(g) is equal to 1/K ✓✓ OR ... less (smaller) than K [3]
Activity 4
The industrial preparation of hydrogen gas is represented by the equation below:
CH4(g) + H2O(g) ⇌ CO(g) + 3H2(g) ∆H > 0
The reaction reaches equilibrium at 1 000 °C in a closed container.
- State Le Châtelier’s principle. (2)
- How will an increase in pressure at 1 000 °C (by decreasing the volume) affect the yield of hydrogen gas? Write down only INCREASES, DECREASES OR NO EFFECT. Explain the answer. (2)
- Give TWO reasons why high temperatures are used for this reaction. (2) [6]
hint
There are various ways to state Le Châtelier’s Principle correctly.
Solution
- When the equilibrium in a closed system is disturbed the system will shift the equilibrium position (OR re-instate a new equilibrium)✓ as to favour the reaction that will oppose OR cancel OR counteract the change OR disturbance. ✓
OR
When a stress / change is placed on a system in equilibrium the system shifts the equilibrium ( position) OR re-instate a new equilibrium ✓ so as to remove OR cancel OR oppose the stress / change. ✓ (2)
OR
When the conditions affecting an equilibrium are changed, the equilibrium (position) shifts in such a way ✓ as to oppose the change OR cancel the change. ✓ (any two) - Decreases ✓ When the pressure is increased, the reverse reaction is favoured.✓
OR
The reaction that produced the smaller volume/amount of gas is favoured. ✓
OR
4 mol or volumes of gas produces ✓ 2 mol or volumes of gas. ✓ (2) (any two) - Products form at faster rate. ✓
Higher yield of products. ✓ (2) [6]
hint
In physical chemistry, saturation is the point at which a solution of a substance can dissolve no more of that substance. Additional amounts of the solute will appear as a separate phase (usually as a precipitate solid). This point of maximum concentration, the saturation point, depends on the temperature and pressure of the solution as well as the chemical nature of the substances involved.
Activity 5
1. The following hypothetical reaction reaches equilibrium in a closed container at a certain temperature:
X2(g) + Y2(g) ⇋ 2XY(g) ∆H < 0
Which ONE of the following changes will increase the AMOUNT of XY(g)?
- Decrease in temperature
- Increase in temperature
- Increase in pressure
- D. Decrease in pressure (2)
2. The equation below represents a chemical reaction at equilibrium in a closed container.
H2(g) + I2(g) ⇌ 2HI(g) ∆H < 0
Which ONE of the following changes will increase the yield of HI(g) in the above reaction?
- Increase the temperature
- Decrease the temperature
- Increase the pressure by decreasing the volume
- Decrease the pressure by increasing the volume (2)
3. A chemical reaction reaches equilibrium. Which ONE of the following statements regarding this equilibrium is TRUE?
- The concentrations of the individual reactants and products are constant.
- The concentrations of the individual reactants and products are equal.
- The concentrations of the individual reactants are zero.
- The concentrations of the individual products increase until the reaction stops. (2)
4. The reaction represented by the equation below reaches equilibrium.
2CrO24−(aq) + 2H+(aq) ⇌ Cr2O27−(aq) + H2O(ℓ)
yellow orange
Which ONE of the following changes to the reaction mixture will change its colour from yellow to orange?
- Add a catalyst.
- Add water to the reaction mixture.
- Add a few drops of sodium hydroxide solution to the reaction mixture.
- Add a few drops of concentrated hydrochloric acid to the reaction mixture. (2)
5. Give one word for the following phrase:
The stage reached in a reversible chemical reaction when the rate of the forward reaction is equal to the rate of the reverse reaction. (2) [10]
Solution
1. A ✓✓ (2)
2. B✓✓ (2)
3. A ✓✓(2)
4. D ✓✓ (2)
5. (Dynamic/Chemical) equilibrium ✓✓ (2) [10]
NB
Solids and pure liquids are omitted from the Kc expression as their concentration is [1], as multiplying by 1 has no effect.
Activity 6
Write Kc expressions for each of the following reactions:
- N2(g) + 3H2(g) ⇋ 2NH3(g) (1)
- CaCO3(s) ⇋ CaO(s) + CO2(g) (1)
- P4(s) + 6Cℓ2(g) ⇋ 4PCℓ3(ℓ) (1) [3]
Solution
- Kc = [NH3]3
[N2]2.[H2]3 - Kc = [CO2]
- Kc = 1
[CL2]6
Activity 7
The thermal decomposition of calcium carbonate (CaCO 3) is an example of a heterogeneous equilibrium. The decomposition that takes place in a closed container can be represented by the following equation:
CaCO3(s) ⇌ CaO(s) + CO2(g)
Initially 5 g of CaCO3(s) is placed in a closed 500 cm3 container and then heated. Equilibrium is reached at 900°C.
- Why is the above decomposition referred to as a heterogeneous equilibrium? (1)
- Calculate the mass of unreacted CaCO3(s) that remains in the container at equilibrium if Kc for the reaction is 0,0108 at 900°C. (19)
- It is found that the value of Kc increases when the container is heated. Is the forward reaction exothermic or endothermic? Use Le Châtelier’s principle to explain your answer. (5)
- The volume of the container is now decreased to 250 cm3 while the temperature is kept constant. How will each of the following be affected? Give a reason for your answer.
4.1 The value of Kc.
4.2 The number of mol CaCO3(s) present in the equilibrium mixture. (10)
4.3 The concentration of CO2(g) at the new equilibrium. - More CaCO3(s) is now added to the equilibrium mixture in the 500 cm3 container. How will this change influence the number of moles of CO2(g) in the equilibrium mixture? Give a reason for your answer. (2) [37]
- hint; Refer back to the definition of a heterogeneous equilibrium
Solutions
- Reactants and products are in different phases. 3 (1)
- This answer takes six steps. Answers for questions 3.3 to 3.5 appear below these steps.
Step 1:
Convert the mass of the reactant to moles. ONLY moles may be used in the table.
- M(CaCO3) = 40 + 12 + 3(16) = 100 g·mol–1
n = m = 5 = 0,05 mol
M 100
Step 2:
Write the expression for Kc and substitute all the known values – remember that the value of Kc was given.
- Kc = [CO2]
0,0108 = [CO2]
∴ [CO2] = 0,0108 mol·dm–3
Step 3:
Use the calculated CO2(g) concentration and the formula for concentration (c = n/V ) to calculate the number of moles of CO2 in the equilibrium mixture. The volume of the container (500 cm3) was given. Convert the volume to dm3. Show all working.
- c = n ∴ 0,0108 = n ∴ n = (0,5)(0,0108) = 0,0054 mol CO2
V 0,5
Step 4:
Find the column with 2 values. Complete the mol used or produced line for this compound. (0 mol CO2 initially + produced mol CO2 = 0,0054 mol
∴ mol CO2 produced = 0,0054 mol)
Step 5:
From the balanced equation ratio 1 : 1 : 1 determine the ratio of the moles used and produced. Therefore 1 : 1 : 1 becomes 0,0054 (from Step 3): 0,0054 : 0,0054
Step 6
For the reactant CaCO3 ... initial mol – mol used = final mol at equilibrium
∴0,05 mol – 0,0054 = final mol at equilibrium = 0,0046 mol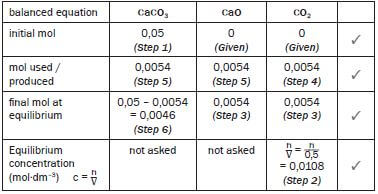
∴0,0046 mol CaCO3 remains unreacted at equilibrium
M(CaCO3) = 40 + 12 + 3(16) = 100 g·mol–1
- n = m ∴ 0,0046 = m ∴ m = (0,0046)(10) = 0,46 g
M 100
∴ 0,46g CaCO3 remains unreacted. (19)
Hint : Mass was asked, so convert the mol calculated to mass n = m
M
3. Endothermic. ✓
- Kc increases ∴ the forward reaction or reaction to the right is favoured. ✓✓
- According to Le Châtelier’s principle, an increase in temperature ✓ the forward reaction or will favour the endothermic reaction reaction to the right must be endothermic. ✓ (5)
4.1 Remains the same. Only temperature affects the value of Kc.
4.2 Increases. ✓✓
- Decreasing the volume increases the pressure. According to Le Châtelier’s Principle, when the pressure is increased, the reaction which decreases the pressure by producing fewer moles of gas products, is favoured. ✓
As 0 mol (g) ∴ 1 mol (g), the reverse reaction is favoured. ✓ - 4.3 Remains the same. ✓✓
Kc = [CO2]. The temperature remains constant ∴ Kc remains constant ∴ the concentration of CO2 will remain constant. ✓ (10)
5. Remains the same. Adding a solid does not affect the equilibrium, it does not change the concentration of a reagent. ✓✓ (2) [37]
Activity 8
NB:
- When∆H > 0 it means the reaction to the right (the forward reaction) is endothermic
- When∆H < 0 the forward reaction is exothermic
hint
- These are the initial concentrations – before the new equilibrium is established. V = 1 dm3 (given)
∴the values of the initial concentrations are equal to the initial number of moles.
c= n and c = n
V 1
∴ n = c(1)
The equation below represents an equilibrium reaction in a sealed 1dm3 container.
NO2(g) + NO(g) ⇋ N2O(g) + O2(g) ΔH > 0
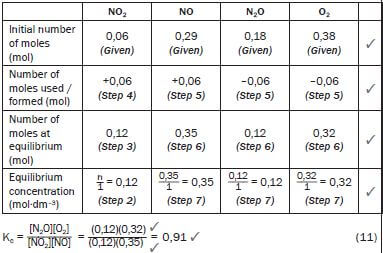
Equilibrium was reached at a certain temperature and the value of Kc was 3,93. The concentration (in mol·dm–3) of each reactant and product in the container at equilibrium was:
- [NO2] = 0,06 mol·dm–3
- [NO] = 0,29 mol·dm–3
- [N2O] = 0,18 mol·dm–3
- [O2] = 0,38 mol·dm–3
One of the conditions affecting the equilibrium is changed and a new equilibrium is established. At the new equilibrium, the concentration of the NO2(g) is 0,12mol·dm–3.
1. Calculate the Kc value at the new equilibrium. (11)
2. Which condition, concentration or temperature, was changed? (1)
3. Give an explanation for the answer to Question 4.2. (1) [13]
Solutions
- NO2(g) + NO(g) + Energy ⇌ N2O(g) + O2(g)
∆ [NO2] = [NO2]final − [NO2]initial = (0,12 – 0,06) = 0,06 mol NO2 is formed. but NO2 is the product of the reverse reaction
∴ the reverse reaction was favoured.From the balanced reaction equation, 1 NO2 : 1 NO : 1 N2O : 1 O2
∴0,06 mol N2O + 0,06 mol O2 → 0,06 mol NO2 + 0,06 mol NO
So at equilibrium:
Temperature.
Kc was initially 3,93 (given) now it is 0,91. If the Kc value changed, the temperature must have changed – only a change in temperature can change the value of Kc. (1)
Activity 9
Consider the following reaction equation for the production of ammonia in the Haber process.
NB
- The Haber Process is the name of the chemical process to manufacture NH3
N2(g) + 3H2(g) ⇌ 2NH3(g) ∆H < 0
In each of the following cases:
- State how Kc changes and
- Briefly explain your answer.
1. The concentration of the nitrogen gas is increased. (1)
2. The pressure in the reaction system is decreased. (1)
3. FeO is added to the reaction system. (1)
4. The reaction system is heated. (5) [8]
Solutions
1. Remains constant because... ✓ (1)
2. ... Kc is only affected by a change in temperature. ✓ (1)
3. Kc Decreases ✓ (1)
4. Heating the reaction system favours the endothermic, reverse reaction ✓ which decreases the temperature ✓ of the system ∴ the concentration of the forward product, [NH3] ✓ decreases while the concentrations of the forward reactants, [N2] and [H2] increase ✓ ∴ Kc decreases. ✓ (5) [8]
Activity 10
The reaction represented by the equation below reaches equilibrium.
Co(H2O)62+(aq) + 4Cℓ–(aq) ⇋ CoCℓ42–(aq) + 6H2O(ℓ) ΔH>0
pink blue
Which ONE of the following changes to the reaction mixture will changes its colour from blue to pink?
- Add a catalyst
- Place the reaction mixture in a container with hot water.
- Add a few drops of concentrated hydrochloric acid to the reaction mixture.
- D. Add water to the reaction mixture. (2) [2]
Solution
1. D [2]
Activity 11
0,25 mol of A and 0,15 mol of B are introduced into a 1 dm3 vessel. A and B react and reach an equilibrium which can be represented by the following equation: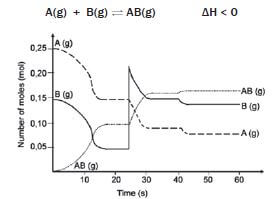
hint: Make use of the graph to read off the values
The graph shows the number of moles of A(g), B(g) and AB(g) vs time, under varying conditions.
- What are the concentrations of A(g), B(g) and AB(g) at t = 20 s? (3)
- How can we be sure that the system has reached equilibrium at t = 20 s? (1)
- 3. Give an explanation for the change that occurs at t = 25 s. (1)
- Explain the change in concentrations of A(g) and B(g) between t = 25 s and t = 35 s. (3)
- How does the equilibrium constant at t = 35 s compare with the equilibrium constant at t = 25 s? (No calculation is required.) (1)
- Assuming that the volume was kept constant, what was done at t = 40 s that produced the change as shown? Explain your answer. (5)
- Do any reactions occur during the interval t = 30 s and t = 40 s? Briefly explain your answer. (4) [18]
Solutions
- [A] = 0,15 mol·dm–3 [B] = 0,05 mol·dm–3 [AB] = 0,1 mol·dm–3 (3)
- The number of mol of A, B and AB remains constant from 16 s to 25 s. ✓(1)
- The amount of B is increased by adding B to the system, thus increasing the [B]. ✓(1)
- With the addition of B, [B] increases and the forward reaction is favoured ✓ according to Le Châtelier’s principle, so that some of B reacts with A to form more AB. Hence the [A] and [B] 3 decrease and [AB] increases. ✓ (3)
- Kc remains constant provided the temperature remains constant. ✓ (1)
- The temperature was lowered. ✓ It was not a concentration change, because there are no peaks at 25 s ✓. Therefore it must be a temperature change. From the graph, it can be seen that the forward reaction was favoured ✓ because there is an increase in the number of moles of AB, and a decrease in the number of moles of A and B when establishing the new equilibrium. ✓ According to the information given, the forward reaction is exothermic. ✓ According to Le Châtelier’s Principle, a decrease in temperature favours the exothermic reaction. (5)
- Yes, but since equilibrium has been reached, ✓ the forward and reverse reactions take place at the same time ✓ and at the same ✓ rate, so that no changes occur in the concentrations of A, B or AB. ✓ (4) [18]
Activity 12
Nitrogen and oxygen gases react in a sealed container according to the following equation:
O2(g) + N2(g) ⇋ 2NO(g)
After the reaction reaches equilibrium, certain changes are made. The following graph of rate of reaction versus time represents the situation.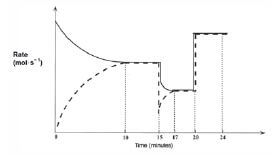
- Write the equation for the reaction represented by the dashed line on this graph. (2)
- What is represented by the section of the graph between the 10th and 15th minute? Explain your answer. (2)
- A temperature change takes place at t = 15 minutes.
3.1 Was the temperature increased or decreased at t = 15 minutes? (1)
3.2 Explain whether the forward reaction is exothermic or endothermic. (3)
3.3 What effect does this temperature change have on the equilibrium constant (Kc)? Explain your answer. (3) - A pressure change is introduced at t = 20 minutes.
4.1 Was the pressure increased or decreased? (3)
4.2 Explain how this change in pressure affects the amounts of each gas at equilibrium. (2) [16]
Solutions
- 2NO(g) → N2(g) + O2(g) ✓✓ (2)
- The system is in equilibrium ✓ because the rates of the forward and reverse reactions are equal. ✓ (2)
- 3.1 Decreased. ✓ (1)
3.2 The rates of both the forward and the reverse reactions decrease ✓, but the rate of the reverse reaction decreases more. ✓ The rate of the endothermic reaction will always decrease the most when a reaction mixture is cooled. Thus the reverse reaction is endothermic. That means that the forward reaction must be exothermic and was favoured by the decrease in temperature. ✓
O2(g) + N2(g) ⇌ 2NO(g) + Energy (3)
3.3 Increases. ✓
Since Kc = [NO]2
[N2][O2] (3)
and the forward reaction is favoured ✓ which causes an increase in [NO], the value of Kc increases. ✓ (3) - 4.1 Increased. ✓ The pressure is probably caused by a volume decrease. This results in the concentrations (c = n/V ) of the reactants, as well as the products, to increase ✓ , which in turn causes an equal increase in the forward and reverse reaction rates. ✓ (3)
4.2 The amount (n) of reactants and products does not change, ✓ since 2 moles of gaseous reactant molecules ⇌ 2 moles of gaseous product molecules. ✓ (2) [16]
Activity 13
A fertiliser company produces ammonia on a large scale at a temperature of 450°C. The balanced equation below represents the reaction that takes place in a sealed container.
N2(g) + 3H2(g) ⇌ 2NH3(g) ∆H < 0
To meet an increased demand for fertiliser, the management of the company instructs their engineer to make the necessary adjustments to increase the yield of ammonia. In a trial run on a small scale in the laboratory, the engineer makes adjustments to the TEMPERATURE, PRESSURE and CONCENTRATION of the equilibrium mixture. The graphs below represent the results obtained.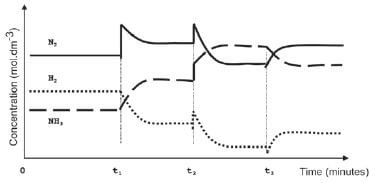
- Identify the changes made to the equilibrium mixture at each of the following times:
1.1 t1 (2)
1.2 t2 (2)
1.3 t3 (2) - At which of the above time(s) did the change made to the reaction mixture lead to a higher yield of ammonia? Write down only t1 and/or t2 and/or t3 (2)
- The engineer now injects 5 mol N2 and 5 mol H2 into a 5 dm3 sealed empty container. Equilibrium is reached at 450 °C. Upon analysis of the equilibrium mixture, he finds that the mass of NH3 is 20,4 g. Calculate the value of the equilibrium constant (Kc) at 450 °C (9) [17]
Solutions
- 1.1 The concentration of nitrogen ✓ is increased ✓ / More Nitrogen ✓ added ✓ (any two) (2)
1.2 The pressure ✓ on the system is increased ✓ (2)
1.3 The temperature ✓ is increased ✓ (2) - t1 3and t2 ✓ (2)
- Pay special attention:
This is how marks are allocated to the answer of this Kc calculation:- Use of n = m/M ✓
- n(NH3) at equilibrium = 1,2 mol ✓
- Using ratio n(N2) : n(H2) : n(NH3) = 1 : 3 : 2 ✓
- n(N2) at equilibrium (initial - change) ✓
- n(H2) at equilibrium (initial - change) ✓
- divide by volume (calculation of concentration) ✓
- Kc expression ✓
- Substitution into Kc expression ✓
- Final answer ✓ (9)
 [17]
[17]
Activity 14
2 mol of NO2(g) and an unknown amount of N2O4(g) are sealed in a 2 dm3 container, that is fitted with a plunger, at a certain temperature. The following reaction takes place:
2NO2(g) ⇌ N2O4(g)
At equilibrium it is found that the NO2 concentration is 0,4 mol·dm–3. The equilibrium constant at this temperature is 2.
- Calculate the initial amount (in mol) of N2O4(g) that was sealed in the container. (8)
The plunger is now pushed into the container causing the pressure of the enclosed gas to increase by decreasing the volume - How will this change influence the amount of nitrogen dioxide at equilibrium? Only write down INCREASES, DECREASES or REMAINS THE SAME. (1)
- Use Le Châtelier’s principle to explain your answer to QUESTION 2.1.2. (2) [11]
Solutions
1.
| 2NO2 | N2O4 | |
| Initial number of mole (mol) | 2 | x |
| Number of moles used/formed (mol) | -1,2 | +0,6 |
| Number of moles at equilibrium(mol) | 0,8 | x + 0,6 |
| Equilibrium concentration (mol·dm -3) | 0,4 | (x + 0,6) 2 |
- Kc = [N2O4]
[NO2]2
2 = (x + 0,6)2
(0,4)2
x = 0,004 mol
2. Decreases (1)
3. When the pressure is increased the system will try to decrease the pressure. The forward reaction (2 mol to 1 mol) is favoured. (2) [11]
Activity 15
Name the industrial process for the production of ammonia (1) [1]
Solution
- Haber (Bosch) process [1]
Activity 16
Sulphuric acid is an important substance used in the manufacture of fertilisers. The equation below represents one of the steps in the industrial preparation of sulphuric acid.
2SO2(g) + O2(g) ⇋ 2SO3(g) ∆H < 0
- Write down the name of the process used to prepare sulphuric acid in industry. (1)
- Write down the NAME or FORMULA of the catalyst used in the process in question (1)
- Is the forward reaction exothermic or endothermic? Give a reason for the answer. The reaction reaches equilibrium at a certain temperature in a 2 dm3 closed container. On analysis of the equilibrium mixture, it is found that 0,6 mol of SO2(g), 0,5 mol of O2(g) and 0,4 mol of SO3(g) are present in the container. (1)
- List THREE changes that can be made to this equilibrium to increase the yield of SO3(g) (3)
- The temperature is NOW increased and the reaction is allowed to reach equilibrium for the second time at the new temperature. On analysis of this new equilibrium mixture, it is found that 0,2 mol of for this reaction at the new temperature. (8) [14]
Solutions
- Contact process ✓ (1)
- V2O5 (vanadium pentoxide) ✓ (1)
- Exothermic∆H < 0 ✓ (1)
- Any three: ✓✓ ✓
Decrease temperature
Increase pressure
Increase concentration of both/any one of the reactants
Remove SO3 continuously (3) -
Kc = [SO3]2 = (0,1)2 = 0,21SO2 O2 SO3 Molar ratio 2 1 2 Initial quantity (mol) 0,6 0,5 0,4 Change (mol) 0,2 0,1 0,2 Quantity at equilibrium 0,8 0,6 0,2 Concentration (mol·dm–3) 0,4 0,3 0,1
[SO2]2[O2] (0,4)2(0,3)
NB
This how marks are allocated to the answer of this Kc calculation:
- Change in n(SO3) = 0,2 mol ✓
- Ratio n(SO2) : n(O2) : n(SO3) = 2 : 1 : 2 ✓
- n(SO2) at equilibrium = initial + change ✓
- n(O2) at equilibrium = initial + change ✓
- divide three equilibrium amounts by 2 (calculation of concentration) ✓
- Kc expression ✓
- Substitution into Kc expression ✓
- Final answer ✓ (8) [14]
Activity 17
The reaction below represents the catalysed step in the contact process:
2SO2(g) + O2(g) ⇋ 2SO3(g) ∆H <0
The reaction takes place in a closed container and reaches equilibrium at 427°C. How will a HIGHER temperature affect each of the following? Write down only INCREASES, DECREASES or REMAINS THE SAME.
- The rate of production of SO3(g) (2)
- The yield of SO3(g) (2)
- The reaction is investigated on a small scale in the laboratory. Initially 4 mol of SO2(g) and an unknown mass, x, of O2(g) are sealed in a 2 dm–3 flask and allowed to reach equilibrium at a certain temperature. At equilibrium it is found that the concentration of SO3(g) present in the flask is 1,5 mol·dm–3. Calculate the mass of O2(g) initially present in the flask if the equilibrium constant (Kc) at this temperature is 4,5. (8) [12]
Solutions
1. Increases ✓✓ (2)
2. Decreases ✓✓ (2)
3.
| SO2 | O2 | SO3 | |
| Molar ratio | 2 | 1 | 2 |
| Initial quantity (mol) | 4 | x 32 | 0 |
| Change (mol) | 3 | 1,5 | 3 ratio |
| Quantity at equilibrium | 1 | x - 1,5 32 | 3 |
| Concentration (mol ·dm –3 | c = n = 1 = 0,5 V 2 | (x - 48) 64 | 3/2 = 1,5 (divide by 2) |
 (8) [12]
(8) [12]